What is the MHC?
The major histocompatibility complex (MHC) is a collection of glycoproteins embedded in the plasma membrane that enable self-recognition of cells as well as the adaptive immune response against pathogens or tumors. MHC bind peptide fragments produced during protein turnover and display them on the cell surface for recognition by T cells. These peptides are normally derived from native cellular proteins, but during infection foreign peptides are also produced and displayed due to pathogen-based metabolism within the host cell. When these foreign peptides are displayed by the MHC, they trigger an immune response, and depending on the type of pathogen and/or its cellular localization, this can lead to the death of a virus-infected cell, activation of macrophages to kill bacteria living in their intracellular vesicles, or B cell activation for the production of antibodies that eliminate or neutralize extracellular pathogens. Self-recognition does not result in cell death in a normal immune response.
A large repertoire of MHC molecules provide broad protection against infection because the genes are 1) polygenic, i.e., every individual contains a set of MHC genes that ranges in peptide-binding specificity, 2) highly polymorphic, i.e., there are multiple variants of each gene within the population, and 3) MHC expression is codominant, i.e., both sets of alleles function.
The MHC is known as Human Leukocyte Antigen (HLA) in humans and H-2 in mouse. Three classes of protein are encoded; MHC class I and II are involved in antigen presentation. Class III proteins do not present antigens and are structurally and functionally poorly defined.
MHC Class I
Class I MHC are found on the cell surface of all mammalian nucleated cells as well as platelets, where they display peptide fragments from within the cell to cytotoxic CD8+ T lymphocytes (CTLs). Peptides generated by cytosolic proteosome degradation identify the cell as “self”. While pathogen-derived peptides instigate programed cell death by apoptosis. Class I MHC can also activate cell death by natural killer cells, when normal surface class I MHC levels are reduced by certain viruses and tumors.
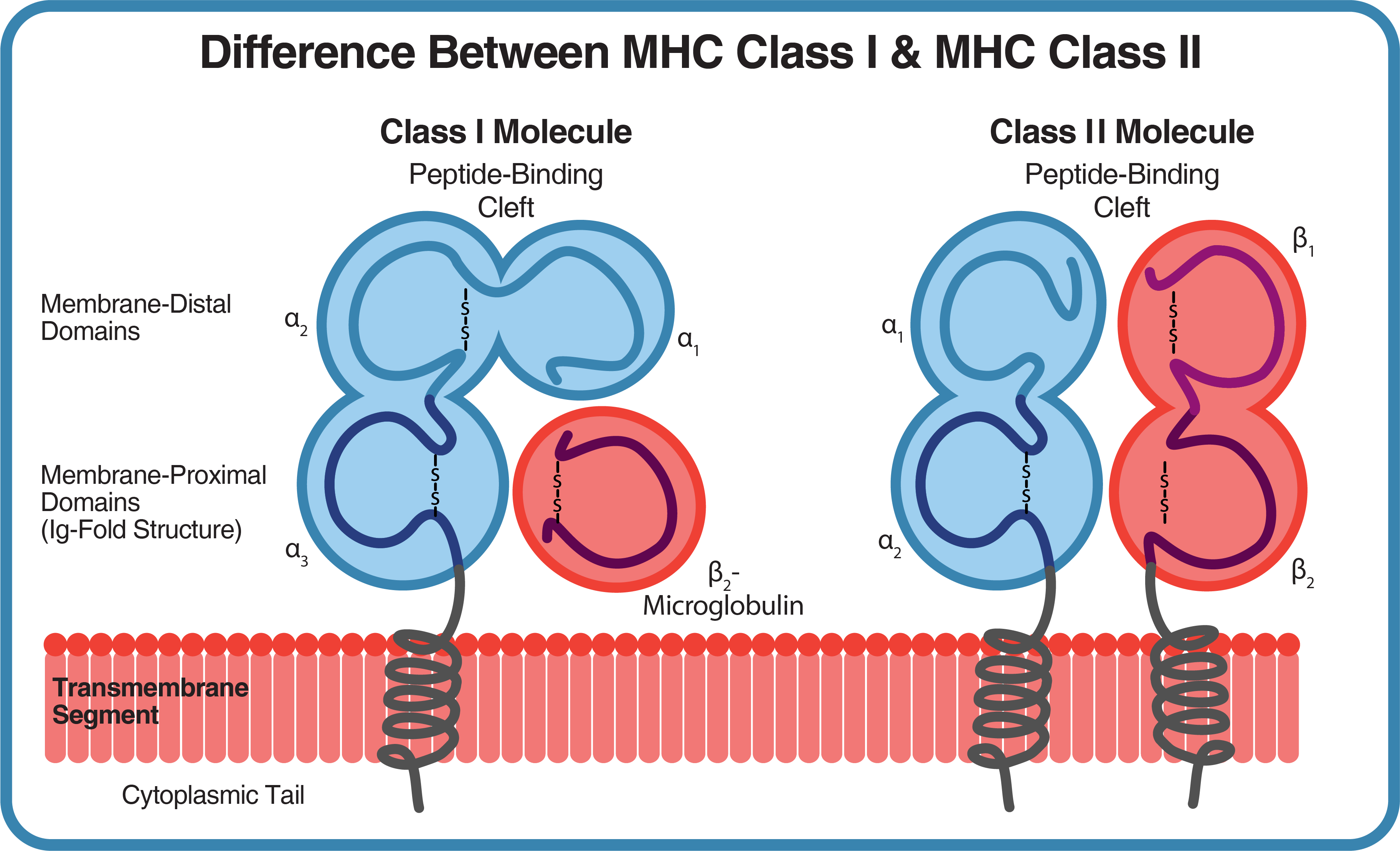
MHC class I molecules are transmembrane heterodimers that consist of two noncovalently linked polypeptide chains: α and β2-microglobulin. Only the α chain is polymorphic and encoded by the MHC. The first two domains of α (α1 and α2) create the peptide binding groove, where the antigen peptide is presented on the extracellular surface, while the α3 domain associates with the CD8 co-receptor of CTLs. This CD8-MHC interaction holds the MHC class I molecule in place while the T cell receptor (TCR) checks the coupled peptide for antigenicity. If the TCR detects a foreign epitope, then apoptosis is triggered.
In humans, the three main MHC class I genes are HLA-A, -B, and -C, with alleles designated as e.g., HLA-A*0201. In mouse, the genes are H2-K, -D, and -L, with haplotypes given further letter designations.
MHC Class II
Class II MHC are found on antigen-presenting cells (APC), such as B cells, mononuclear phagocytes, macrophages, dendritic cells as well as some endothelial cells. MHC class II molecules present peptides derived from extracellular proteins, including extracellular pathogens, to CD4+ T helper cells. The extracellular protein is endocytosed, digested in lysosomes to peptide fragments which are loaded onto MHC class II molecules, and then migrates to the cell surface where it attracts a naïve helper T cell. The CD4 molecule docks to the APC’s MHC class II molecule and the TCR binds the epitope presented, activating the naïve T cell and initiating an immune response when the peptide is identified as foreign.
Class II MHC are heterodimers of an α and β subunit; the genes for which are all polymorphic and encoded by the MHC locus. Two α helixes and a β sheet create an antigen binding groove that is open at both ends and can accommodate a longer antigen than MHC class I molecules, expanding the variety of antigenic peptides capable of presentation.
In humans, MHC class II proteins are encoded by six isotypes: HLA-DM, HLA-DO, HLA-DP, HLA-DQ, and HLA-DR, with A or B designations for the α- or β-chain and numerical designations for alleles. In mouse, two immune response molecules, I-A and I-E, are encoded by multiple isotypes.
Leinco’s MHC Antibody Products
Human MHC Antibody Products
A variety of MHC class I and II antibodies are available. W6/32 (anti-human HLA-A, B, C; MHC Class I) is available with different conjugations. BB7.6 (anti-human HLA A2, B7; MHC Class I) is available in PLATINUM and GOLD Functional Grades. L243 (anti-human HLA-DR; MHC Class II) is available with various conjugations and at PLATINUM and GOLD Functional Grades. B7/21 (anti-human HLA-DP; MHC class II) is available for flow cytometry and at GOLD Functional Grade for in vivo experiments. 1a3 (anti-human HLA-DQ; MHC class II) is available for a variety of analyses and at PLATINUM and GOLD Functional Grades.
| Target | Clone | Specificity |
| MHC-I | W6/32 | HLA-A,B,C |
| MHC-I | BB7.6 | HLA A2, B7 |
| MHC-II | L243 | HLA-DR |
| MHC-II | B7/21 | HLA-DP |
| MHC-II | 1a3 | HLA-DQ |
Mouse MHC Antibody Products
Antibodies have been generated against a variety of mouse MHC proteins. These include clones against the class I H-2Kb (AF6-88.5, Y-3) and H-2Kd (A4C8.1-Do9) that have a variety of conjugations for experimental purposes or are PLATINUM or GOLD Functional Grade as well as a clone against class II I-A/I-E (M5/114/15/2).
| Target | Clone | Specificity |
| MHC-I | A4C8.1-Do9 | H-2Kd |
| MHC-I | AF6-88.5 | H-2Kb |
| MHC-I | Y-3 | H-2Kb |
| MHC-I | M1/42.3.9.8 | H-2 |
| MHC-I | SF1.1.10 Coming Soon | H-2Kd |
| MHC-I | 15-3-1S | H-2Kk, H-2Dk |
| MHC-I | 34-1-2S Coming Soon | H-2Kd, H-2Dd |
| MHC-I | 28-14-8S | H-2Db |
| MHC-I | D2C5-Ky82 | H-2b, H-2p, H-2q, H-2w16 |
| MHC-II | Y-3P | I-A |
| MHC-II | M5/114.15.2 | I-A/I-E |
| MHC-II | 14-4-4S | I-Ek/RT1-D |
| MHC-II | 10-3.6.2 | I-Ak, I-Ar, I-Af, I-As, I-Ag7 |

