Immune cells play critical roles in the defense of the body against infectious agents and the development and spread of cancer. The immune system involves two primary lines of defense against non-self pathogens. The innate immune system is the first line of defense that quickly responds in a broad way. This involves natural killer cells, macrophages, neutrophils, dendritic cells, mast cells, basophils, and eosinophils. The adaptive immune system takes over if needed and specifically targets and remembers the pathogen to provide long-lasting immunity. The adaptive immune system includes T lymphocytes (T cells), B lymphocytes (B cells), and antibodies.
Tissues and Organs Involved
Many cells work together as part of the innate and adaptive immune system.
One of the primary components of the immune system is the bone marrow, which produces both T and B lymphocytes. While B cells will remain in the bone marrow to mature, T cells continue to another organ, the thymus.
The thymus is a specialized primary lymphoid organ where T cells develop and mature, including the acquisition of antigen receptors such as CD4 and CD8, and differentiate into helper T cells and cytotoxic T cells.
Mature T and B lymphocytes move to other tissues and organs of the immune system, the lymph nodes and spleen, in preparation for activation. The spleen filters the blood, where macrophages engulf and digest cellular debris, pathogens, and other foreign substances, and aids the immune system by identifying pathogenic microorganisms.
Immune Cell Types
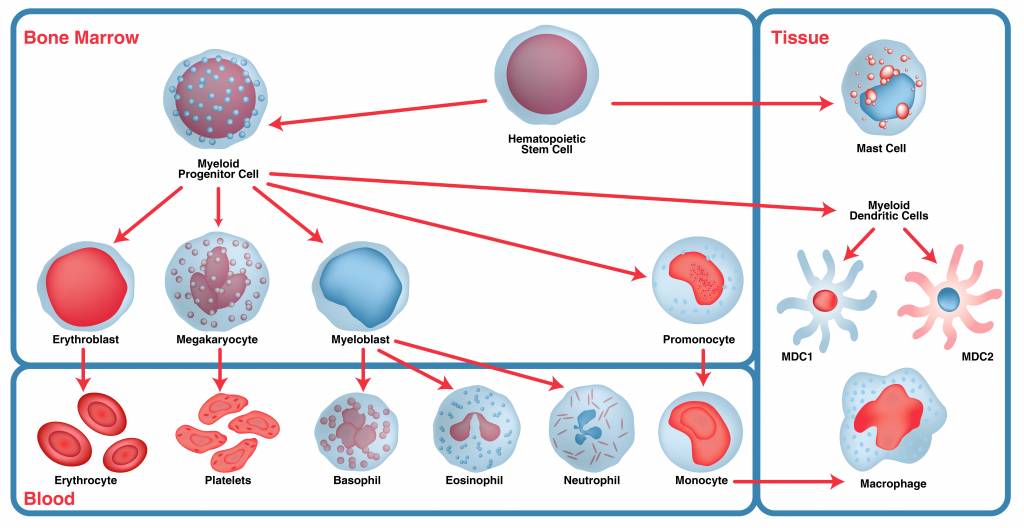
Immune cells are derived from blood stem cells through two lineages: myeloid stem cells and lymphoid stem cells.
Myeloid Lineage Cells
- Granulocytes are leukocytes of the innate immune system that have enzyme containing granules in their cytoplasm.
- Neutrophils are the most abundant innate immune cells. As first responders, they ingest harmful foreign particles, bacteria, and dead or dying cells in the blood, and also modulate inflammation by various mechanisms.
- Eosinophils help in killing parasites, tumor cells, and immune system regulation.
- Basophils travel to the site of infection and release histamines when damaged, which contributes to the inflammatory response, and prostaglandins that help increase blood flow to the infection site.
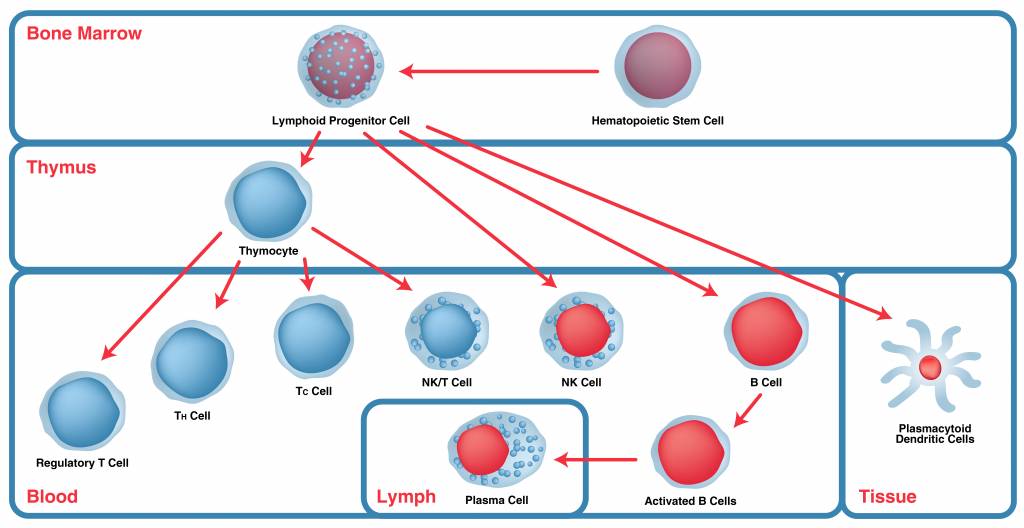
Lymphoid Lineage Cells
- T cells have several roles and are divided into CD8+ T cells or CD4+ T cells depending on which cluster of differentiation (CD) protein is present on the cell surface.
- CD8+ T cells (cytotoxic T cells, cytotoxic lymphocytes, or CTLs) recognize and eliminate virus-infected cells and cancer cells.
- CD4+ T cells are divided into subsets:
- T-helper (TH)1 cells coordinate immune responses against intracellular microbes.
- TH2 cells coordinate immune responses against extracellular pathogens such as parasites by alerting B cells, granulocytes, and mast cells.
- TH17 cells produce interleukin 17 (IL-17) that activates immune and nonimmune cells, and they recruit neutrophils.
- Tregs (regulatory T cells) monitor and inhibit the activity of other T cells.
- Killer T cells (cytotoxic T cells) are the primary effector cells of adaptive immunity. They can attach to and kill cancer cells.
- B cells make antibodies and can also internalize antigens and present them to T cells. Memory B cells are long lived and remember previous exposure to antigens. Plasma B cells are activated B cells, each making one specific antibody in significant quantity.
- Natural killer (NK) cells are cytotoxic lymphocytes that are critical to the innate immune system. They respond rapidly to infection by viruses and other intracellular pathogens, and tumor formation without the need for antibodies and major histocompatibility complex (MHC).
- Natural killer T (NKT) cells are a heterogeneous group of T cells that share properties of both T cells and natural killer cells.
Monocytes
Monocytes are produced in the bone marrow and move into tissues throughout the body to differentiate into macrophages and dendritic cells.
Macrophages
Macrophages engulf and digest pathogens by phagocytosis. The macrophages identify the pathogens, which can include cancer cells, microbes, cellular debris, and foreign substances by the presence of nonself surface proteins that distinguish the pathogen from healthy body cells. Macrophages play a critical role in innate immunity and also help initiate specific defense mechanisms (adaptive immunity) by
recruiting other immune cells such as lymphocytes. They can increase inflammation but also have an anti-inflammatory role through the release of cytokines.
Dendritic Cells
Dendritic cells (DCs) are specialized antigen-presenting cells (APCs) that form an interface between innate and adaptive immunity. Once activated, DCs migrate to the lymph nodes and interact with T cells and B cells to initiate and shape the adaptive immune response.
DCs present endogenous and exogenous antigens to T cells in the context of MHC molecules, resulting in antigen tolerance or priming and triggering of an effector T cell response. DCs also help maintain the immune memory of B cells, including the production of cytokines and other factors that promote B cell activation and differentiation.
Expected Levels of Immune Cells in Blood
| Level (x 109/L) | |||
| Cell Type | Adult | Pediatric (4-7 years old) | Neonate (0-1 days old) |
| WBC | 3.6–10.6 | 5.0–17.0 | 9.0–37.0 |
| Neutrophils | 1.7–7.5 | 1.5–11.0 | 3.7–30.0 |
| Lymphocytes | 1.0–3.2 | 1.5–11.1 | 1.6–14.1 |
| Monocytes | 0.1–1.3 | 0.1–1.9 | 0.1–4.4 |
| Eosinophils | 0.0–0.3 | 0.0-0.7 | 0.0–1.5 |
| Basophils | 0.0–0.2 | 0.0–0.3 | 0.0–0.7 |
Source: Modified from Wikipedia.
Complement System — A Non-cellular Part of the Immune System
The complement system, or cascade, enhances (complements) the ability of antibodies and phagocytic cells to clear microbes and damaged cells from an organism, promote inflammation, and attack the cell membranes of pathogens. Complement components in circulation are synthesized by the liver while complement components in tissues are mainly synthesized by macrophages.
The complement system comprises several related proteins that form a self-amplifying proteolytic cascade. Products later in the cascade can create pores in cell membranes, leading to cell lysis, while many of the proteolytic fragments generated during the cascade have other useful biological activities. The complement cascade can be activated by an antibody first binding to a particular antigen, direct
binding to microbial surfaces, or by circulating proteins of the innate system that bind to pathogenassociated molecular patterns (PAMPs).
The Role of Immune Cells in Infectious Diseases
The immune response depends on the type of microorganism together with the nature of the pathogenicity (extracellular, intracellular, toxin-based, etc.) and virulence level or, in the case of parasites, the life cycle. For example:
Bacteria
- Neutralizing antibodies are synthesized by the B cells if the bacterial pathogenicity is due to a toxin.
- Opsonizing antibodies destroy extracellular bacteria.
- The complement system is activated, especially by gram-negative bacterial lipid layers.
- Phagocytes kill most bacteria.
- CD8+ T cells can kill cells infected by intracellular bacteria.
Fungi
- The innate immunity includes defensins and phagocytes.
- CD4+ T helper cells are responsible for the adaptive immune response against fungi.
- Dendritic cells secrete IL-12 after ingesting fungi, and IL-12 activates the synthesis of gamma interferon which activates the cell-mediated immunity
Viruses
- Interferon, NK cells, and phagocytes prevent the early spread of viruses.
- Specific antibodies and complement proteins neutralize viruses and can limit spread and reinfection.
- The adaptive immunity is most important in protecting against viral infection, including CD8+ T cells that kill viruses and CD4+ T cells that dominate the effector cell population.
Parasites
- Parasitic infection stimulates various mechanisms of immunity due to their complex life cycle.
- Both CD4+ and CD8+ T cells protect against parasites.
- Macrophages, eosinophils, neutrophils, and platelets can kill protozoa and worms.
- Eosinophils and the stimulation of IgE by Th2 CD4+ T cells are involved in killing intestinal worms.
- Inflammatory responses also combat parasitic infections.
The Role of the Immune System in Cancer
Immune cells play key roles in both tumor proliferation and elimination. Chronic inflammation is a necessary consequence of cancer progression that includes genomic and epigenomic instability, immune evasion, angiogenesis, and metastasis. Chronic inflammation can also be a primary cause of cancer.
An aberrant innate and adaptive immune response contributes to tumorigenesis by selecting for aggressive cells, inducing immunosuppression, and stimulating cancer cell proliferation and metastasis. During early tumor development, cytotoxic immune cells such as natural killer (NK) cells and CD8+ T cells recognize and eliminate the more immunogenic cancer cells.
Selected cancer cells that survive progress to clinically detectable tumors and adopt different strategies of peripheral immune tolerance and recruitment of immunosuppressive immune cells, such as regulatory T cells, pro-tumoral macrophages, and pro-metastatic neutrophils that can subdue other tumoricidal cells.
Macrophages are important drivers of chronic cancer-associated inflammation and are involved in every step of cancer progression, from early neoplastic transformation throughout metastatic progression to therapy resistance.
Processes such as angiogenesis, extracellular matrix (ECM) remodeling, and immune evasion are mediated by tumor-associated macrophages (TAMs), tumor-associated neutrophils (TANs), and immature dendritic cells (DCs), resulting in rapid tumor progression and metastasis.
In contrast, cytotoxic macrophages and neutrophils, natural killer (NK) cells, and mature DCs eliminate tumor cells in primary sites and after dissemination.
T cells are the second most frequent immune cell type found in human tumors besides TAMs and modulate immunity and direct inflammatory or anti-inflammatory processes. CD8+ T cells are the most prominent anti-tumor cells. After being primed by APCs, the CD8+ T cells differentiate into cytotoxic T lymphocytes (CTLs) that attack the tumor.
The constant selective pressure of the effector response results in the selection of tumor variants that escape immune recognition and enter a phase of outgrowth that is not blocked by effector immune cells. At the same time, the tumor induces the recruitment of regulatory CD4+ T cells (Tregs) that counteract anti-tumor immune cells by diverse mechanisms. Tumors with high Treg infiltration have the
worst prognosis.
Immunophenotyping
Immunophenotyping is a powerful tool that uses flow cytometry to gain insight into the composition and dynamics of cell populations in an immune response. The technique involves identifying cell types in heterogeneous cell populations by using different fluorescently-tagged antibodies that target various CD markers. Different combinations of antibodies can be used to identify different groups and sub-groups of cells. For example, CD3 can be used as a general marker for T cells followed by other CD markers to identify regulatory T cells (CD4 and CD25), T helper cells (CD4), and cytotoxic T cells (CD8). Immunophenotyping can be used for discovery and can also be applied in clinical settings, for example to help diagnose and classify a leukemia or lymphoma, or refine the analysis of cancer cell lines by determining the presence or absence of cancer cell markers that correlate with different degrees of severity.
Antibodies as Tools in Immunophenotyping
Leinco Technologies provides a range of antibodies that can be used in immunophenotyping in research (see tables).
Myeloid
| Monoclonal Abs | ||||||||||
| Target | Alternative Name | Polyclonal | Unconjugated | FITC | PE | APC | PerCP | DyLight® | Reactivity | Clone |
| Stem and Progenitor Cells | ||||||||||
| CD117 | c-Kit; SCF-R | S579 | Human | |||||||
| Erythroblast | ||||||||||
| CD71 | Transferrin Receptor | C163 | C165 | C328 | Available | Human | T56/14 | |||
| C275 | C277 | C278 | C355 | C1933 | Mouse | R17217 | ||||
| Megakaryocyte and Platelets | ||||||||||
| CD61 | Integrin β3 | C191 | C193 | C194 | C327 | C1822 | Available | Human | F11 | |
| Human | PM6/13 | |||||||||
| Granulocyte: Basophil | ||||||||||
| CD123 | IL3RA | I-651 | Human | 32703 | ||||||
| I-719 | Mouse | 151231 | ||||||||
| Granulocyte: Eosinophil | ||||||||||
| CD44 | C183 | C185 | C186 | C320 | C1804 | Available | Human | E1/2 | ||
| Human | F10-44-2 | |||||||||
| C251 | C253 | C254 | C349 | C1894 | Available | Human/Mouse | IM7 | |||
| Granulocyte: Neutrophil | ||||||||||
| CD15 | C157 | C159 | C160 | C316 | C2015 | Available | Human | TG1 | ||
| CD16 | C6850 | C6854 | C6851 | C6852 | C6853 | Available | Human | 3G8 | ||
| Human | GRM1 | |||||||||
| C247 | C249 | C250 | C348 | Available | Mouse | 2.4G2 | ||||
| Monocyte | ||||||||||
| CD11b | C133 | C135 | C136 | C307 | C1727 | Available | Human | ICRF44 | ||
| C227 | C229 | C230 | C341 | C1906 | Available | Mouse | M1/70 | |||
| CD14 | C153 | C155 | C156 | C315 | C1867 | Available | Human | UCHM-1 | ||
| Myeloid Dendritic cells: MDC2 | ||||||||||
| CD141 | Thrombomodulin | T537 | T547 | Human | 501733 | |||||
Lymphoid
| Monoclonal Abs | ||||||||||
| Target | Alternative Name | Polyclonal | Unconjugated | FITC | PE | APC | PerCP | DyLight® | Reactivity | Clone |
| Stem and Progenitor Cells | ||||||||||
| CD117 | c-Kit; SCF-R | S579 | Human | |||||||
| T lymphocytes (T Cells) | ||||||||||
| CD2 | C109 | C111 | C112 | C308 | C1839 | Available | Human | G11 | ||
| CD3 | C105 | C107 | C108 | C309 | C2033 | Available | Human | UCHT-1 | ||
| C207 | C2839 | C1737 | Available | Mouse | 500A2 | |||||
| C1757 | C2234 | C2037 | C2036 | Available | Mouse | 145-2C11 | ||||
| T lymphocytes (Cytotoxic) | ||||||||||
| CD8 | C117 | C119 | C120 | C312 | C2068 | Available | Human | UCHT-4 | ||
| C297 | C299 | C300 | C301 | C1664 | Available | Mouse | 2.43 | |||
| C215 | C217 | C218 | C339 | C1746 | Available | Mouse | 53-6.7 | |||
| T lymphocytes (Helper) | ||||||||||
| CD4 | C9210 | Human | OKT-4 | |||||||
| C211 | C213 | C214 | C338 | C1840 | Available | Mouse | GK1.5 | |||
| T lymphocyte (Regulatory) | ||||||||||
| CD4 | See Above | See Above | See Above | See Above | See Above | See Above | See Above | See Above | ||
| CD25 | IL2Rα | LT300 | LT304 | LT303 | Available | Human | HU107 | |||
| C1189 | C1191 | C1192 | C1193 | C1924 | Available | Mouse | PC61 | |||
| T lymphocyte (NK/T) | ||||||||||
| CD3 | See Above | See Above | See Above | See Above | See Above | See Above | See Above | See Above | ||
| T lymphocyte (Activated) | ||||||||||
| CD25 | IL2Rα | See Above | See Above | See Above | See Above | See Above | See Above | See Above | See Above | |
| CD30 | C1384 | Human | 81337 | |||||||
| B lymphocyte (B cell) | ||||||||||
| CD19 | C141 | C143 | C144 | C317 | C1978 | Available | Human | SJ25-C1 | ||
| C2088 | C2105 | C2107 | C2106 | C2108 | Available | Mouse | 1D3 | |||
| CD20 | C1602 | C1654 | C1895 | C1896 | C1682 | Available | Human | 2H7 | ||
| B lymphocyte (Activated) | ||||||||||
| CD19 | See Above | See Above | See Above | See Above | See Above | See Above | See Above | See Above | ||
| CD25 | IL2Rα | See Above | See Above | See Above | See Above | See Above | See Above | See Above | See Above | |
| CD30 | See Above | See Above | See Above | See Above | See Above | See Above | See Above | See Above | ||
| Natural Killer (NK) Cell | ||||||||||
| CD56 | NCAM | See Above | See Above | See Above | See Above | See Above | See Above | See Above | See Above | |
Find Out More
- Gonzalez H, Hagerling C, Werb Z. Roles of the immune system in cancer: from tumor initiation to metastatic progression. Genes Dev. 2018 Oct 1;32(19-20):1267-1284. doi: 10.1101/gad.314617.118. PMID: 30275043; PMCID: PMC6169832.
- Balakrishnan A, Vig M, Dubey S. Role of myeloid cells in the tumor microenvironment. J Cancer. Metastasis Treat 2022;8:27. https://dx.doi.org/10.20517/2394-4722.2022.33
- Alexandra C. Costa, Joana M.O. Santos, Rui M. Gil da Costa, Rui Medeiros, Impact of immune cells on the hallmarks of cancer: A literature review, Critical Reviews in Oncology/Hematology, Volume 168, 2021, 103541, ISSN 1040-8428, https://doi.org/10.1016/j.critrevonc.2021.103541. (https://www.sciencedirect.com/science/article/pii/S1040842821003280)
- Nardy AF, Freire-de-Lima CG, Morrot A. Immune responses to infectious diseases. https://austinpublishinggroup.com/ebooks/innovative-immunology/chapters/II-14-12.pdf

