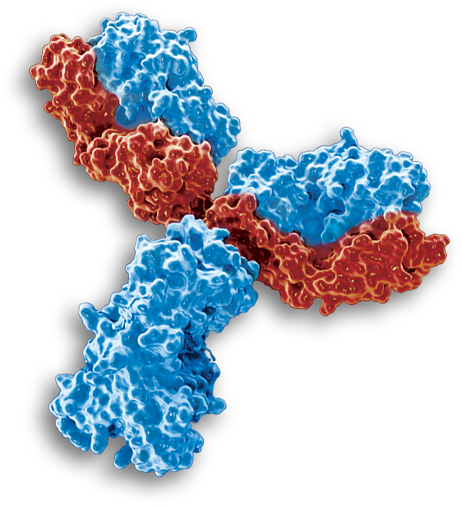
Leinco Technologies is a leader in the development and manufacturing of recombinant antibodies for early discovery research and diagnostics. We offer an extensive line of recombinant biosimilar antibodies at the in vivo functional purity levels you have come to know and trust from Leinco Technologies. Recombinant biosimilar antibodies are designated for research use only and may not be used for therapeutics. Our biosimilars are made available to researchers to avoid purchasing therapeutic antibodies at pharmaceutical prices for important groundbreaking experiments. Learn more about Leinco’s industry leading purity specifications, current product offerings and custom services.
Don’t see the conjugate you need? Click the button below to request a conjugate!
Categories
Filters