Recombinant Human Protein CTLA-4 (CD152)
Recombinant Human Protein CTLA-4 (CD152)
Product No.: C1319
Alternate Names CD152, Cytotoxic T Lymphocyte-Associated Antigen-4, Ly-56, CELIAC3, GSE, IDDM12, CELIAC3, GSE, IDDM12 Product Type Recombinant Protein Expression Host sf Insect Cells Species Human |
Data
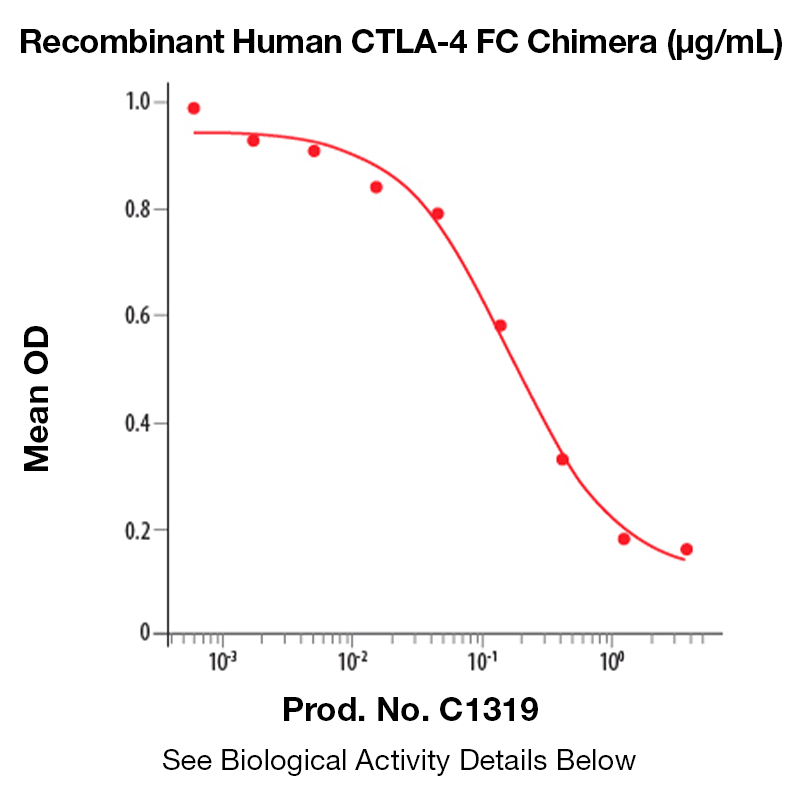
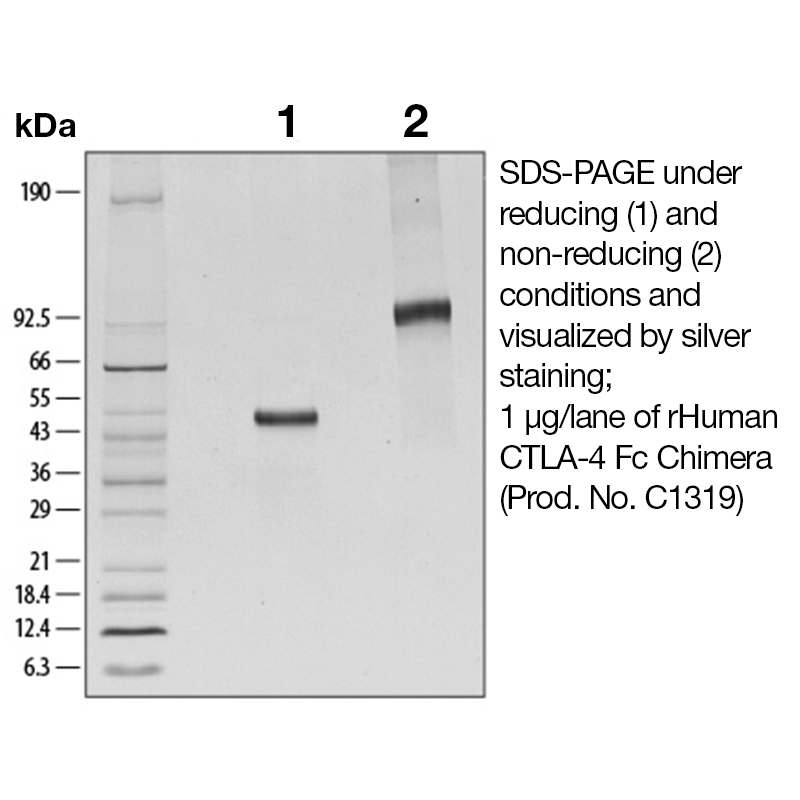
BackgroundCTLA-4 is a 33 kD member of the Ig superfamily similar to CD28 in amino acid sequence, structure, and genomic organization. CTLA-4 is a protein receptor that functions as an immune checkpoint and downregulates immune responses. It is involved in the development of protective immunity and thymocyte regulation, in addition to the induction and maintenance of immunological tolerance. CTLA-4 has therapeutic potential both as an agonist to reduce immune activity, and an antagonist to increase immune activity. Protein DetailsPurity >90% by SDS-PAGE and analyzed by silver stain. Endotoxin Level <0.1 EU/µg as determined by the LAL method Biological Activity The biological activity of Human CTLA-4 was determined by its ability to inhibit IL-2 production by Jurkat cells stimulated with Raji cells or 1 μg/ml rhB7-1/Fc in the presence of PHA (Linsley, P.S. et al., 1991, J. Exp. Med. 174:561). The expected ED<sub>50</sub> is typically 2 - 4 μg/ml with Raji cells or 0.1 - 0.4 μg/ml with rhB7-H1/Fc. Fusion Protein Tag Fc Fusion Protein Protein Accession No. Amino Acid Sequence amhv aqpavvlass rgiasfvcey aspgkatevr vtvlrqadsq vtevcaatym mgneltfldd sictgtssgn qvnltiqglr amdtglyick velmypppyy lgigngtqiy vidpepcpds dfiegrmdpk scdkthtcpp cpapellggp svflfppkpk dtlmisrtpe vtcvvvdvsh edpevkfnwy vdgvevhnak tkpreeqyns tyrvvsvltv lhqdwlngke ykckvsnkal papiektisk akgqprepqv ytlppsrdel tknqvsltcl vkgfypsdia vewesngqpe nnykttppvl dsdgsfflys kltvdksrwq qgnvfscsvm healhnhytq kslslspgkh hhhhh
N-terminal Sequence Analysis Ala37 State of Matter Lyophilized Predicted Molecular Mass The predicted molecular weight of Recombinant Human CTLA-4 is Mr 40 kDa. Predicted Molecular Mass 40 Formulation This recombinant protein was 0.2 µm filtered and lyophilized from modified Dulbecco’s phosphate buffered saline (1X PBS) pH 7.2 – 7.3 with no calcium, magnesium, or preservatives. Storage and Stability This lyophilized protein is stable for six to twelve months when stored desiccated at -20°C to -70°C. After aseptic reconstitution, this protein may be stored at 2°C to 8°C for one month or at -20°C to -70°C in a manual defrost freezer. Avoid Repeated Freeze Thaw Cycles. See Product Insert for exact lot specific storage instructions. Country of Origin USA Shipping Next Day Ambient NCBI Gene Bank Leinco Protein AdvisorPowered by AI: AI is experimental and still learning how to provide the best assistance. It may occasionally generate incorrect or incomplete responses. Please do not rely solely on its recommendations when making purchasing decisions or designing experiments. Recombinant Human Protein CTLA-4 (CD152) is a critical tool for research applications focused on immune regulation, immunotherapy, and T-cell biology. It enables precise investigation of immune checkpoint mechanisms, therapeutic development, and functional assays relevant to cancer, autoimmunity, and tolerance. Key scientific reasons to use recombinant human CTLA-4 in research include:
Summary of applications:
Using recombinant human CTLA-4 provides a standardized, reproducible reagent for these applications, facilitating high-quality, translational research in immunology and related fields. Yes, recombinant human CTLA-4 (CD152) protein can be used as a standard for quantification and calibration in ELISA assays. This is a well-established application supported by multiple validated protocols and quality-controlled products. Suitability for ELISA StandardsRecombinant human CTLA-4 protein is specifically designed and validated for use as an ELISA standard. The protein is quality control tested by ELISA assay for each production lot, ensuring consistency and reliability across experiments. These standards are guaranteed suitable for sandwich ELISA applications, which is the most common format for CTLA-4 quantification. Key Specifications for CalibrationWhen using recombinant CTLA-4 as a standard, consider these important characteristics: Protein Properties: Recombinant CTLA-4 proteins are typically expressed in mammalian systems (such as human 293 cells or CHO cells) and contain affinity tags (His tag, Twin-Strep tag, or Fc tag) for purification and detection. The protein exhibits high purity, generally exceeding 85-95% as determined by SDS-PAGE. Molecular Weight Considerations: The calculated molecular weight of CTLA-4 is approximately 16.5 kDa, though the protein may migrate at 20-30 kDa under reducing conditions due to glycosylation. This is important to account for when preparing your standard curve. Binding Characteristics: Recombinant CTLA-4 protein demonstrates specific binding to B7-2 (CD86) and can be captured by anti-CTLA-4 antibodies with defined linear ranges (typically 1-20 ng/mL depending on the specific protein variant). Practical Considerations for Standard PreparationWhen reconstituting lyophilized recombinant CTLA-4 standards, follow the manufacturer's protocol carefully. Standards are typically reconstituted in appropriate dilution buffers to achieve a stock concentration (commonly 10 ng/mL). Use a fresh standard for each assay to ensure accuracy. The assay range for human CTLA-4 detection typically spans from approximately 23.4 pg/mL to 1500 pg/mL, depending on the specific kit formulation, providing adequate dynamic range for most quantification applications. Recombinant Human Protein CTLA-4 (CD152) has been validated for a range of applications in published research, primarily in the fields of immunology, cancer immunotherapy, and autoimmune disease studies. Key validated applications include:
Summary Table of Validated Applications
These applications are supported by a substantial body of published research and ongoing clinical trials, reflecting the central role of recombinant CTLA-4 in both basic and translational immunology. To reconstitute and prepare Recombinant Human Protein CTLA-4 (CD152) for cell culture experiments, first centrifuge the vial briefly to collect the lyophilized powder at the bottom. Add sterile distilled water to achieve a final concentration typically between 0.1–1.0 mg/mL; a common working concentration is 0.2–0.5 mg/mL. Avoid vigorous mixing—gently swirl or invert the vial to dissolve the protein. Detailed protocol:
Additional notes:
Summary of best practices:
This protocol ensures optimal solubility and activity of recombinant CTLA-4 for cell culture experiments. References & Citations1. Layrisse, Z. et al. (2009) Hum Immunol.70(7):532-5 2. Mak, TW. et al. (1995) Science 270: 985 3. Delneste, Y. et al. (1999) Eur. J. Immunol 29: 3596 4. Vijayakrishnan, L. et al. (2004) Immunity 20: 563 Certificate of AnalysisIMPORTANT Use lot specific datasheet for all technical information pertaining to this recombinant protein. |
Related Products
Prod No. | Description |
|---|---|
C1748 | |
C2445 | |
C1614 | |
C2855 | |
C2860 | |
C2856 | |
C399 | |
C1319 | |
C1340 | |
C400 |
 Products are for research use only. Not for use in diagnostic or therapeutic procedures.
Products are for research use only. Not for use in diagnostic or therapeutic procedures.