Anti-Rhinovirus 16 [Clone R16-7]
Anti-Rhinovirus 16 [Clone R16-7]
Product No.: 18758
- -
- -
Clone R16-7 Target Rhinovirus 16 Formats AvailableView All Product Type Monoclonal Isotype Mouse IgG2b Applications IHC , WB |
Data
- -
- -
Antibody DetailsProduct DetailsReactive Species Rhinovirus 16 Host Species Mouse Immunogen Purified HRV16 Product Concentration Lot Specific Formulation This monoclonal antibody is formulated in phosphate buffered saline (PBS) pH 7.2 - 7.4 with no carrier protein or preservatives added. State of Matter Liquid Product Preparation Antibodies are purified by a multi-step process including the use of protein A or G to assure extremely low levels of endotoxins, leachable protein A or aggregates. Storage and Handling Upon initial thawing, appropriately aliquot and store at -80°C. For long-term storage, keep at -80°C. Avoid repeated freeze-thaw cycles. Country of Origin USA Shipping Next Day 2-8°C Applications and Recommended Usage? Quality Tested by Leinco Immunoblotting: use at 2-5ug/mL.
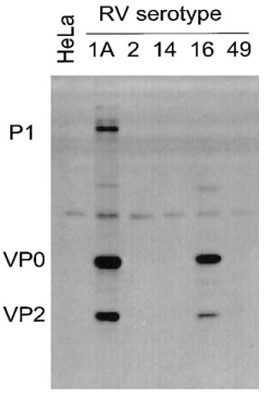
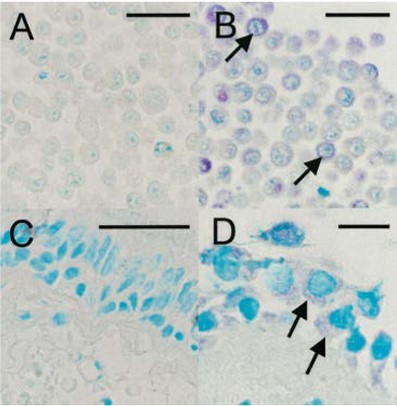
Immunohistochemistry: use at 1-10ug/mL. These are recommended concentrations; Endusers should determine optimal concentrations for their applications. Each investigator should determine their own optimal working dilution for specific applications. See directions on lot specific datasheets, as information may periodically change. DescriptionDescriptionSpecificity This non-neutralizing antibody recognizes capsid protein VP2 (mw ~30kDa) of HRV16, HRV1A, and HRV39 and VP2 precursors VP0 (mw ~37kDa) and P1 (mw ~90kDa). Background Rhinoviruses (HRV), members of the Picornaviridae family, are ubiquitous and highly prevalent RNA viruses. These small, non-enveloped pathogens are characterized by their icosahedral capsid and a single-stranded, positive-sense RNA genome. The viral genome uniquely encodes a single polyprotein that is subsequently cleaved by viral proteases into essential structural and non-structural proteins, vital for viral replication and assembly. With over 100 known serotypes, Human Rhinoviruses are the primary cause of the common cold, leading to millions of upper airway infections annually. Beyond acute symptoms, HRV infections pose a significant health burden by exacerbating lower airway symptoms in vulnerable patient populations. They are well-documented triggers for severe complications in individuals suffering from asthma, cystic fibrosis (CF), and chronic obstructive pulmonary disease (COPD), often leading to increased hospitalizations and diminished quality of life. Our Anti-Rhinovirus 16 monoclonal antibody, Clone R16-7, is specifically developed to target Rhinovirus 16. This highly specific tool is indispensable for researchers investigating HRV pathogenesis, viral diagnostics, and the development of new antiviral therapies or vaccines. Leverage Clone R16-7 for precise detection and characterization of Rhinovirus 16 in your studies. Research Area Infectious Disease References & Citations1) Mosser AG et al. 2002 J Infect Dis 185: 734. 2) Mosser AG et al. 2005 Am J Respir Crit Care Med 171: 645. 3) Jurgeit A et a. 2010 Virology J 7: 264. 4) Chattoraj SS et al. 2011 Infect Immun 79: 4131. Technical ProtocolsCertificate of Analysis |