SARS-CoV-2 Recombinant Proteins
Leinco Technologies understand the importance of quality recombinant proteins and enzymes that produce consistent results for studying COVID-19 and other related diseases. These proteins are vitally important for both vaccine development and diagnostic tests. With over 25 years of experience, Leinco’s SARS-CoV-2 recombinant proteins are produced with > 95% purity level by SDS page, an endotoxin of ≤ 1.0 EU/mg, and contain no carrier protein.
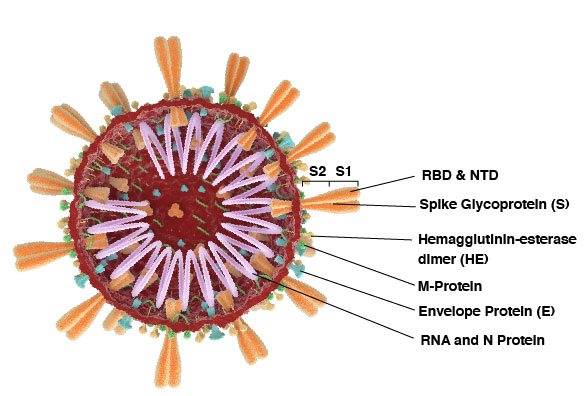
The angiotensin-converting enzyme 2 (ACE2) is the host portal for SARS-CoV-2 entry into cells1. The trimeric spike (S) protein of SARS-CoV-2 is primed by TMPRSS2 and consists of the S1 and S2 domains1. The S1 subunit contains the N-terminal domain (NTD) and the receptor-binding domain (RBD), encoded by all coronaviruses. In addition, the Nucleocapsid (N) Protein is the most abundant protein for coronavirus binding to the RNA genome. The N protein plays a role in transcription, replication, and packaging of the viral RNA genome, while also affecting host cell responses such as cell cycle and translation2.
Multiple SARS-CoV-2 variants have emerged in 2020-2021, while circulating globally. Variants include the United Kingdom (also known as 20I/501Y.V1, VOC 202012/01, or B.1.1.7), South Africa (also known as 20H/501Y.V2 or B.1.351) and Brazil (also known as P.1). Scientific researchers asses if these variants, caused by multiple mutations, have changes in transmissibility, health severity, and the effectiveness of diagnostics, therapeutics and vaccines against them.
Product | Applications | Prod No. | Package Size |
|---|---|---|---|
ELISA | S351 | 1.0 mg ⋅ 100 µg ⋅ 500 µg | |
ELISA | S151 | 1.0 mg ⋅ 100 µg ⋅ 500 µg | |
ELISA | S952 | 1.0 mg ⋅ 100 µg ⋅ 500 µg | |
ELISA | S951 | 1.0 mg ⋅ 100 µg ⋅ 500 µg | |
ELISA | S251 | 1.0 mg ⋅ 100 µg ⋅ 500 µg | |
ELISA | S451 | 1.0 mg ⋅ 100 µg ⋅ 500 µg |
Product | Applications | Prod No. | Package Size |
|---|---|---|---|
ELISA ⋅ WB | S854 | 1.0 mg ⋅ 100 µg ⋅ 500 µg | |
ELISA | S856 | 1.0 mg ⋅ 100 µg ⋅ 500 µg | |
ELISA | S851 | 1.0 mg ⋅ 100 µg ⋅ 500 µg | |
ELISA | S853 | 1.0 mg ⋅ 100 µg ⋅ 500 µg | |
ELISA | S855 | 1.0 mg ⋅ 100 µg ⋅ 500 µg | |
ELISA | S850 | 1.0 mg ⋅ 100 µg ⋅ 500 µg | |
ELISA | S848 | 100 µg ⋅ 500 µg |
- Hoffmann et al., 2020, Cell 181, 271–280
- Kang, S. et al. (2020) Acta Pharm Sin B. Apr 20. doi: 10.1016/j.apsb.2020.04.009.