Recombinant Influenza B NP (B/Florida/4/2006)
Recombinant Influenza B NP (B/Florida/4/2006)
Product No.: F650
- -
- -
Alternate Names Influenza B Virus NP, NP, Nucleocapsid Protein, Nucleoprotein Product Type Recombinant Protein Expression Host HEK-293 Cells Species Influenza B Virus Applications ELISA ⋅ WB |
Data
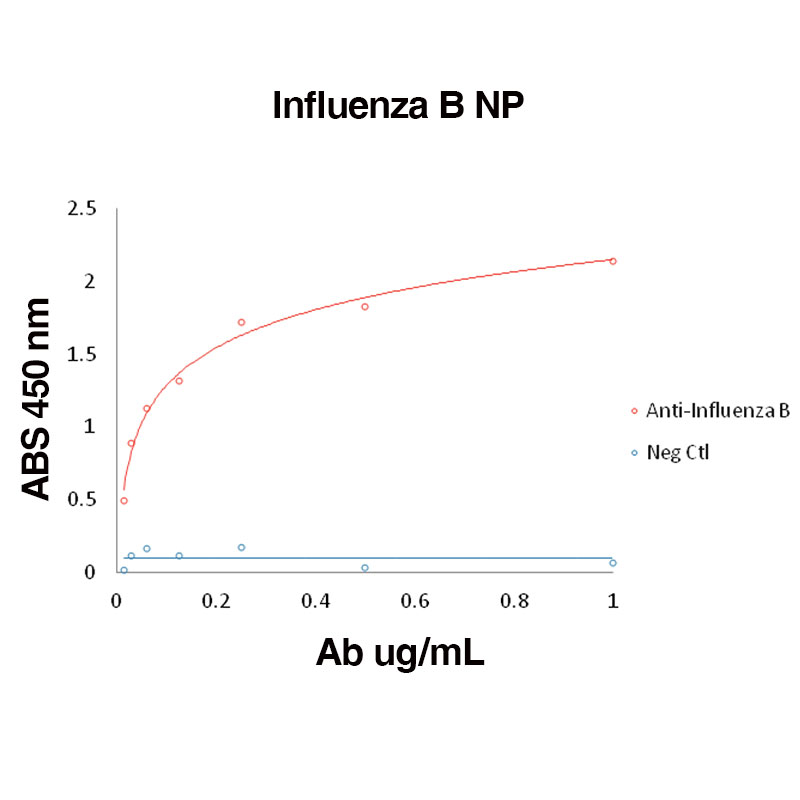 Direct binding of anti-Influenza B antibody to Influenza B NP (Leinco Prod. No.: F650)
Direct binding of anti-Influenza B antibody to Influenza B NP (Leinco Prod. No.: F650)
Binding was measured by ELISA. Recombinant Influenza B NP was immobilized at 1 µg/mL. Anti-Influenza B NP antibody was titrated.
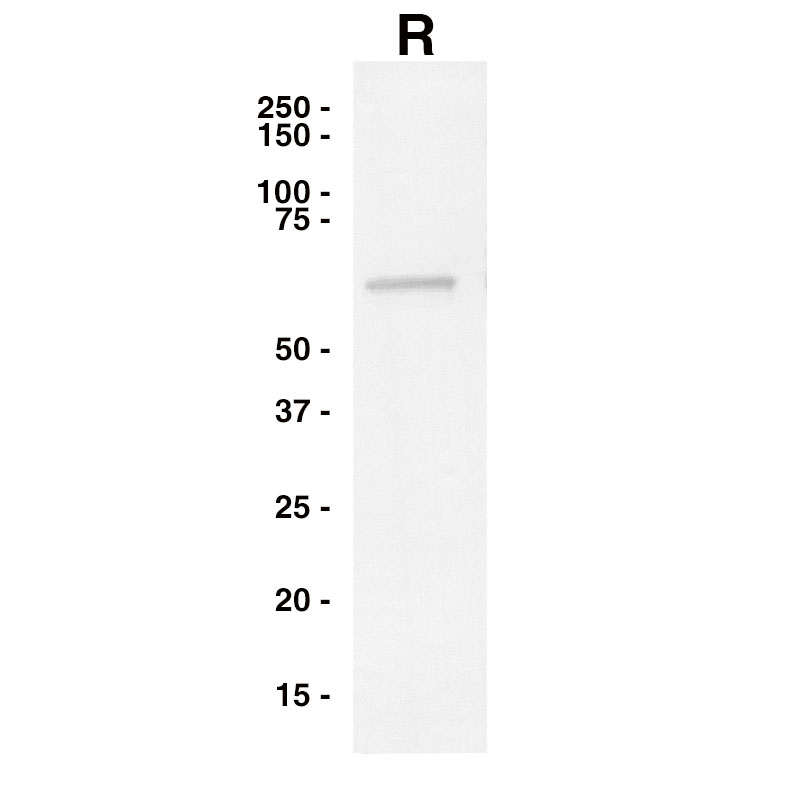 Western Blot
Western Blot
Purified Recombinant Influenza B NP (Leinco Prod. No.: F650) was separated on SDS-PAGE under reducing conditions and probed with an anti-Influenza B NP antibody.
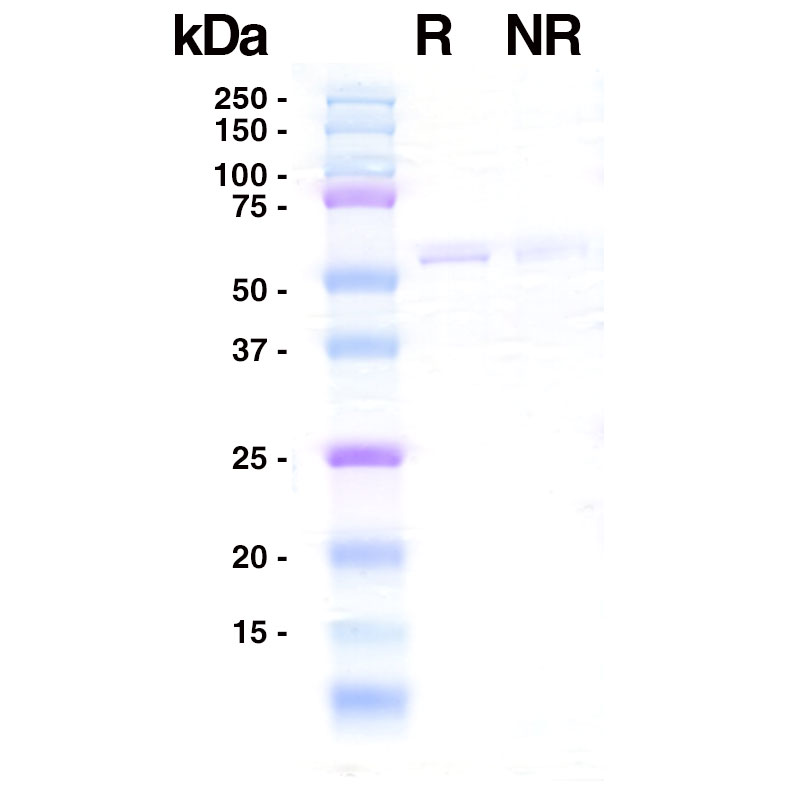 SDS-PAGE
SDS-PAGE
R: 5µg reduced Recombinant Influenza B Nucleocapsid (Leinco Prod. No.: F650).
NR: 5µg non-reduced Recombinant Influenza B Nucleocapsid (Leinco Prod. No.: F650).
- -
- -
BackgroundThere are four types of Influenza (flu) viruses: type A, B, C, and D. Influenza A and B viruses are routinely spread in people and other mammals, while also known for seasonal flu epidemics each year. Influenza B is a Betainfluenzavirus in the virus family Orthomyxoviridae. Influenza B is classified into two distinct lineages, B/Victoria and B/Yamagata1. Influenza B can be even further classified into specific clades (also known as groups) and sub-clades (also known as sub-groups) based on similarity of their HA/NA gene sequences1. The Nucleocapsid protein or nucleoprotein (NP) of influenza virus B negative-strand RNA’s primary function is to encapsulate the virus genome for the purpose of RNA transcription, replication and packaging2. Specifically, NP is the most abundant viral protein in infected cells2, therefore the NP can be and has been used for anti-influenza drug development3 The NP of influenza A and B viruses share up to 38% of their amino acid sequence, indicating region functionality differences at the amino acid level4. Protein DetailsSpecies Viral Format Purified No Carrier Protein Purity ≥95% by SDS Page Product Concentration ≥1.0 mg/ml Endotoxin Level <0.1 EU/µg Biological Activity ELISA binding Protein Accession No. Amino Acid Sequence MSNMDIDGINTGTIDKTPEEITPGTSGTTRPIIRPATLAPPSNKRTRNPSPERATTSSEDDVGRKTQKKQTPTEIKKSVYNMVVKLGEFYNQMMVKAGLNDDMERNLIQNAHAVERILLAATDDKKTEFQKKKNARDVKEGKEEIDHNKTGGTFYKMVRDDKTIYFSPIRITFLKEEVKTMYKTTMGSDGFSGLNHIMIGHSQMNDVCFQRSKALKRVGLDPSLISTFAGSTIPRRSGATGVAIKGGGTLVAEAIRFIGRAMADRGLLRDIKAKTAYEKILLNLKNKCSAPQQKALVDQVIGSRNPGIADIEDLTLLARSMVVVRPSVASKVVLPISIYAKIPQLGFNVEEYSMVGYEAMALYNMATPVSILRMGDDAKDKSQLFFMSCFGAAYEDLRVLSALTGTEFKPRSALKCKGFHVPAKEQVEGMGAALMSIKLQFWAPMTRSGGNEVGGDGGSGQISCSPVFAVERPIALSKQAVRRMLSMNIEGRDADVKGNLLKMMNDSMAKKTSGNAFIGKKMFQISDKNKTNPVEIPIKQTIPNFFFGRDTAEDYDDLDY State of Matter Frozen; PBS no preservatives SDS-Page Molecular Weight 65 kDa Predicted Molecular Mass The predicted molecular mass is ~65 kDa. Predicted Molecular Mass ~65 kDa Formulation This recombinant protein is aseptically packaged and formulated in 0.01 M phosphate buffered saline (PBS) pH 7.2 - 7.4, 150 mM NaCl with no carrier protein, potassium, calcium or preservatives added. Due to inherent biochemical properties of proteins, certain products may be prone to precipitation over time. Precipitation may be removed by aseptic centrifugation and/or filtration. Storage and Stability This recombinant protein is stable for twelve months when stored at -20°C to -70°C. Avoid Repeated Freeze Thaw Cycles. Country of Origin USA Shipping Frozen Dry Ice Ligand/Receptor Ligand Species Viral Regulatory Status Research Use Only NCBI Gene Bank UniProt.org B4UQF0 Applications and Recommended Usage ? (Quality Tested by Leinco) SDS-PAGE, WB, ELISA References & Citations1. Types of Influenza viruses (2019) Center for Disease Control and Prevention. 2. Hu Y, Sneyd H, et al. (2017) Curr Top med Chem. 2017; 17(20): 2271–2285. 3. Portela A, Digard P. (2012) J. Gen Virol. 2002 Apr;83(Pt 4):723-734. 4. Sherry L, Smith M, et al. (2014) Journal of Virology. 2014; 88(21); 12326-12338. DOI https://doi.org/10.1128/JVI.01542-14 Technical ProtocolsCertificate of AnalysisIMPORTANT Use lot specific datasheet for all technical information pertaining to this recombinant protein. |
 Products are for research use only. Not for use in diagnostic or therapeutic procedures.
Products are for research use only. Not for use in diagnostic or therapeutic procedures.


